Outpatient acute seizure management at the level of the general practitioner clinic: a proposed treatment algorithm
Prahlad K Sethi, MD1 Dhrumil Shah, MD1 Anuradha Batra, MD 1 Nitin K Sethi, MD2
1 Department Neurology, Sir Ganga Ram Hospital, New Delhi, India
2 Department of Neurology, New York-Presbyterian Hospital, Weill Cornell Medical Center, New York, NY (U.S.A.)
Seizures beget seizures has been a point of contention over the years. There is some scientific evidence to suggest that each seizure increases the risk for future seizures and that failure to control seizures in a timely fashion can lead to status epilepticus (SE). Status epilepticus is a life threatening neurological emergency which can present as an exacerbation of a pre-existing seizure disorder such as in an epilepsy patient who is non-compliant with his anti-epileptic drug (AED) regimen or as the initial manifestation of a seizure disorder (epilepsy) or as the manifestation of other systemic and cerebral insults. Prolonged seizures are also associated with worse neurological outcomes. With the aim to reduce the time to treatment gap, outpatient treatment of seizures is now been explored. We discuss this approach in relation to the health care system of India.
General practitioners (GPs) also referred to as primary care physicians (PCPs) form the backbone of the Indian health care delivery system. Usually, they are in solo practice working in small clinics (offices) which are ill-equipped to handle medical and surgical emergencies such as seizures and SE.
As awareness about coronary artery disease has increased, GPs now feel comfortable administering aspirin and nitrates before transferring the patient to the hospital. With respect to emergency management of seizures, their knowledge and experience is more limited. If the seizure has stopped, the patient is usually referred to a neurologist. If the patient is actively seizing, the patient is referred to the nearest hospital. The time to treatment gap results in increased morbidity and mortality especially for patients presenting with SE. Unlike developed countries, India lacks a well-organized and responsive 911 type medical emergency system manned by well trained and certified emergency medical technicians (EMTs) and paramedics who can administer life-saving 1st line and 2nd line antiseizure medications such as benzodiazepines (lorazepam, diazepam) and phenytoin or fosphenytoin parenterally en-route to the hospital.
Status epilepticus is a neurological emergency. Early effective treatment of SE results in termination of seizure activity and thereby reduction in cerebral hypoxia and damage. Hence every effort should be made to treat SE at the earliest. In India this goal would be best achieved by initiation of treatment of SE at the GP level. Till recently only intravenous benzodiazepine (diazepam or lorazepam) or rectal diazepam was available for the emergent management of seizures and SE. Now drugs which can be rapidly administered via the intranasal or intramuscular routes are also available such as intranasal midazolam and intramuscular fosphenytoin. Neurocritical Care Society guidelines recommend the administration of benzodiazepines via rectal, intramuscular, intranasal or buccal routes if intravenous or oral administration is not feasible. 1Administration via the above routes has been determined to be quick, easy, safe and to achieve high and consistent blood levels of the active compound. 2
We feel that antiseizure drugs in these formulations can be easily administered by a GP at the clinic without any special expertise or formal training.
Intranasal midazolam: Currently in Indian market 2 midazolam formulations are available, one is MIDACIP (Cipla Pharmaceuticals) and other is MIDASPRAY (Intas Pharmaceuticals). They are both metered spray preparations. Two strengths of MIDACIP are commercially available: (1.25mg and 0.5mg)
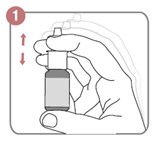
Method of administration of MIDACIP nasal spray (Figure 1):
- Shake the bottle gently.
- Remove the dust cap.
- Hold the bottle with your forefinger and middle finger on either side of the nozzle and your thumb underneath the bottle
- If using first time, spray it six times in the air with the nozzle pointing away from the patient until the consistent mist of the drug is delivered, this is called priming, which ensures that correct dose is delivered.
- If the patient is in supine position, head is slightly lifted upwards and the device should be placed near the patient’s nose.
- Insert the nozzle into patient’s nostril, depress the pump with a firm even stroke. (Patient need not inhale)
- Tilt the patient head backward while spraying, this will avoid swallowing of the solution.
- Administer one spray at a time in each nostril to continue prescribed dose
- Reprime the device for subsequent use if the bottle is not used for more than a day. To reprime spray it two to three times in the air until a fine mist appears. For reusing the device nozzle and dust cap must be washed before storage.
Figure 1. Method of administration of intranasal midazolam.
Each nasal spray delivers either 1.25mg or 0.5mg of midazolam. Dose is titrated according to individual patient weight and full effective dose should be administered. For adults, dose is 5 mg if weight <50 kg and 10 mg if weight>50 kg. The dose should be equally divided and administered into each nostril. For children the recommended dose of MIDACIP nasal spray is 0.2 mg/kg body weight. The dose should be equally divided and administered into each nostril. Placing half the medication in each nostril reduces the volume while doubling the available surface area for absorption.
Table 1. Dosing Guidelines of MIDACIP Nasal Spray in children
| Age (years) | Weight (kg) | Dose (mg) | Metered Doses in Each Nostril |
| ½ – 1 | <10 | 1.25 – 2 | 1 – 2 |
| 1 – 4 | 10 – 16 | 2.5 | 2 – 3 |
| 4 – 10 | 16 – 32 | 5 | 4 – 6 |
| >10 | > 32 | 10 | 10 |
Intramuscular midazolam and fosphenytoin: GPs by virtue of their training can administer intramuscular injections with ease. In India intramuscular midazolam (MIDAZ, Abbott Healthcare or FULSED, Ranbaxy Laboratories) injections are widely available and cost effective. Midazolam is given intramuscularly at the dose of 10mg once or 0.2mg/kg once but not exceeding 10mg. Intramuscular fosphenytoin formulations are also available (Fosolin, Zydus Cadila Healthcare or Fosphen, Intas Pharmaceuticals) but more expensive. These preparations are available as ampoules of 150mg (75mg/ml, 2ml). It is our recommendation that the GP administer 2 ampoules of fosphenytoin stat in the clinic prior to transporting the patient to the hospital. Doing so may abort the seizure and possibly terminate the SE.
Use of the above formulations in the clinic setting by the GP along with established seizure first aid guidelines (Figure 2) has the potential to save many lives and reduce the morbidity from seizures and SE in our country.
Figure 2. Seizure first-aid guidelines (Source: Epilepsy Foundation Eastern Pennsylvania, www.epepa.org)
References
- Brophy GM, Bell R, Claassen J, et al. Guidelines for the evaluation and management of status epilepticus. Neurocrit Care 2012; 17:3–23.
- Agarwal SK, Cloyd JC. Development of benzodiazepines for out-of-hospital management of seizure emergencies. Neurol Clin Pract. 2015; 5:80-85.